
To determine oxidation electrodes, the reduction equation can simply be flipped and its potential changed from positive to negative (and vice versa). The table below is a list of important standard electrode potentials in the reduction state. 
Important Standard Electrode (Reduction) Potentials We could have accomplished the same thing by taking the difference of the reduction potentials, where the absent or doubled negation accounts for the fact that the reverse of the reduction reaction is what actually occurs. Textbook content produced by OpenStax is licensed under a Creative Commons Attribution License. We recommend using aĪuthors: Paul Flowers, Klaus Theopold, Richard Langley, William R. Use the information below to generate a citation. Then you must include on every digital page view the following attribution: If you are redistributing all or part of this book in a digital format, Then you must include on every physical page the following attribution: If you are redistributing all or part of this book in a print format, Want to cite, share, or modify this book? This book uses the The reactions associated with this process are: 
The industrial process typically uses a Downs cell similar to the simplified illustration shown in Figure 17.18. Metallic sodium, Na, and chlorine gas, Cl 2, are used in numerous applications, and their industrial production relies on the large-scale electrolysis of molten sodium chloride, NaCl( l). Concept: An electrochemical cell is composed of an electrolyte and two electrodes, i.e., one anode and one cathode electrode. The Electrolysis of Molten Sodium Chloride To illustrate the essential concepts of electrolysis, a few specific processes will be considered. They move from anode to the cathode in the external circuit. This class experiment can be done by students working either in pairs or threes. 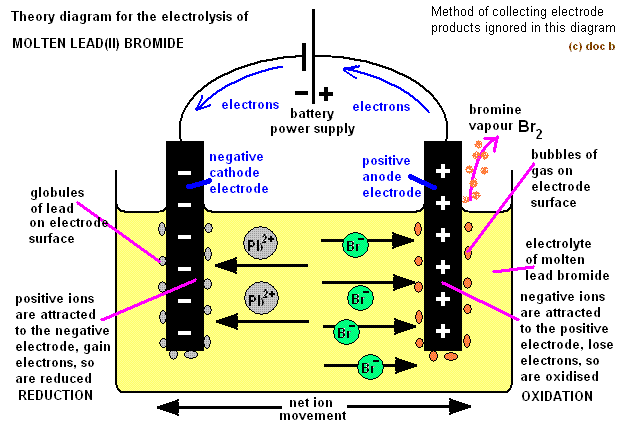
The electrons are supplied by the species getting oxidized. The reaction at the anode is oxidation and that at the cathode is reduction. Here, the anode is positive and cathode is the negative electrode. Perhaps less familiar is the use of electrolysis in the refinement of metallic ores, the manufacture of commodity chemicals, and the electroplating of metallic coatings on various products (e.g., jewelry, utensils, auto parts). The reaction at the anode is oxidation and that at the cathode is reduction. A familiar example of electrolysis is recharging a battery, which involves use of an external power source to drive the spontaneous (discharge) cell reaction in the reverse direction, restoring to some extent the composition of the half-cells and the voltage of the battery. This final section of the chapter will address an alternative scenario in which an external circuit does work on a redox system by imposing a voltage sufficient to drive an otherwise nonspontaneous reaction, a process known as electrolysis. In these cells, electrical work is done by a redox system on its surroundings as electrons produced by the redox reaction are transferred through an external circuit. Perform stoichiometric calculations for electrolytic processesĮlectrochemical cells in which spontaneous redox reactions take place ( galvanic cells) have been the topic of discussion so far in this chapter.Compare the operation of electrolytic cells with that of galvanic cells.Calculate the number of moles of electrons that were transferred. By the end of this section, you will be able to: Calculating the Quantity of Substance Produced or Consumed To determine the quantity of substance either produced or consumed during electrolysis given the time a known current flowed:: Write the balanced half-reactions involved.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
